| From-http://www.ndt-ed.org/EducationResources/CommunityCollege/Materials/Structure/metallic.htm A common characteristic of metallic elements is they contain only one 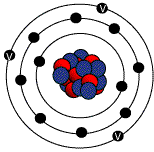 to three electrons in the outer shell. When an element has only one, two or three valence electrons (i.e. electrons in the outer shell), the bond between these electrons and the nucleus is relatively weak. So, for example, when aluminum atoms are grouped together in a block of metal, the outer electrons leave individual atoms to become part of common “electron cloud.” In this arrangement, the valence electrons have considerable mobility and are able to conduct heat and electricity easily. Also, the delocalized nature of the bonds, make it possible for the atoms to slide past each other when the metal is deformed instead of fracturing like glass or other brittle material. to three electrons in the outer shell. When an element has only one, two or three valence electrons (i.e. electrons in the outer shell), the bond between these electrons and the nucleus is relatively weak. So, for example, when aluminum atoms are grouped together in a block of metal, the outer electrons leave individual atoms to become part of common “electron cloud.” In this arrangement, the valence electrons have considerable mobility and are able to conduct heat and electricity easily. Also, the delocalized nature of the bonds, make it possible for the atoms to slide past each other when the metal is deformed instead of fracturing like glass or other brittle material. 
Since the aluminum atoms lose two electrons, they end up having a positive charge and are designated Al3+ ions (cations). These ions repel each other but are held together in the block because the negative electrons are attracted to the positively charged ions. A result of the sharing of electrons is the cations arrange themselves in a regular pattern. This regular pattern of atoms is the crystalline structure of metals. In the crystal lattice, atoms are packed closely together to maximize the strength of the bonds. An actual piece of metal consists of many tiny crystals called grains that touch at grain boundaries. Some Common Features of Materials with Metallic Bonds: - Good electrical and thermal conductors due to their free valence electrons
- Opaque
- Relatively ductile
|

