Questions
Explanations
(1) 1 (2) 5 (3) 9 (4) 14
100 has 2 zeros..pH changes by 2 towards a lower pH
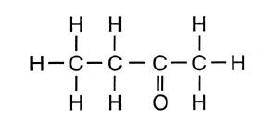
A chemical name for this compound is
(1) butanal (2) butanol
(3) butanone (4) butanoic acid
-one
(1) Small amounts of energy are converted into large amounts of matter.
(2) Small amounts of matter are converted into large amounts of energy.
(3) Heavy nuclei are split into lighter nuclei.
(4) Light nuclei are combined into heavier nuclei.
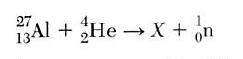
Which particle is represented by X?

atomic number (bottom #) 13 + 2 = x + 0....so 15
atomic # 15 is P
(1) 1/2 (3) 1/8
(2) 1/4 (4) 1/16
1-->1/2/-->1/4-->1/8-->1/16
On to Question 51-53